 As a substitute for chemical separations.Compounds sensitive to temperature rise.Other methods are not feasible: Similar or very small volatilities.6 Equipment for liquid-liquid extraction.In a liquid-liquid extraction operation, the solution to which the components are intended to be separated is called the liquid extraction solvent to be used to separate the desired component, refined to the feed already treated and extract to the solution with the solute. Maguire, Rachael L.The Extraction Liquid -Liquid is, by distillation, the most important basic operation in the separation of homogeneous liquid mixtures. It consists of separating one or several substances dissolved in a solvent by transferring it to another insoluble, or partially insoluble, solvent in the first one. The transfer of matter is achieved through direct contact between the two liquid phases. One of the phases is dispersed in the other to increase the interfacial surface and increase the flow of transferred material. This simulation was made at the University of Colorado Boulder, Department of Chemical and Biological Engineering. This simulation runs on desktop using the free Wolfram Player. In the “view phases” option, the one- and two-phase regions are labeled. The left of the phase envelope boundary is the α phase, and the right is the β phase. Gray tie lines are shown when “show tie lines” is selected, and grid lines are shown when “show grid lines” is selected. These compositions are shown when “show α phase” and “show β phase” are selected. When the black point is in the two-phase region, a tie line (large-dash black line) and its endpoints correspond to the two phases (α, β) in equilibrium. The mass fraction of a component in the mixture is read off the axis that is the same color as that component. 
Each corner of the triangle corresponds to a pure component. 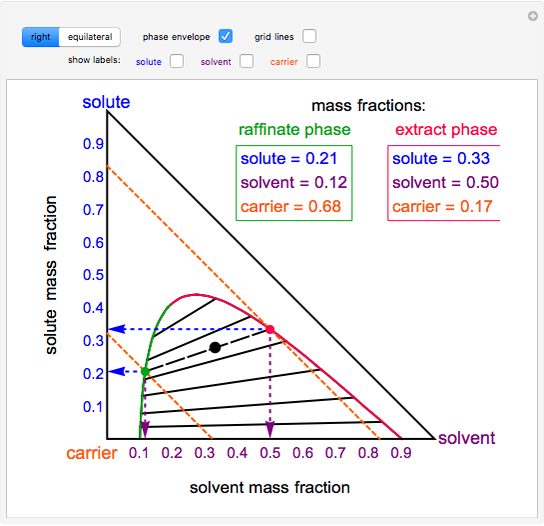
In the “view diagram” option, the black point, which can be moved to any location within the triangle by clicking on that location, represents the overall composition of the mixture. This represents the phase behavior of ternary mixtures that are only partially miscible over a range of compositions so phase separation occurs. 
This simulation shows a ternary phase diagram that has a phase envelope. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
